KFF Health News Experts Weigh In on Crucial Health Policy Debates

KFF Health News continues to be a prominent voice in the national conversation surrounding critical health policy issues, with its leading journalists providing expert analysis on a range of pressing topics. In early May, chief Washington correspondent Julie Rovner and editor-at-large for public health Céline Gounder offered insights on vaccine research, the complex landscape of vaping regulations, access to vital medications, and the far-reaching implications of a significant Supreme Court ruling. Their appearances on prominent national broadcasts underscore KFF Health News’ commitment to informing the public and policymakers alike on matters of public health and healthcare.
Rovner Addresses Federal Vaccine Policy, Vaping, and Drug Access on Science Friday
On May 8, Julie Rovner, a veteran journalist with extensive experience covering federal health policy, joined the popular public radio program Science Friday to dissect several multifaceted federal policy initiatives. Her discussion encompassed the ongoing developments in vaccine research, the evolving regulatory environment for vaping products, and the persistent challenges associated with ensuring equitable access to prescription drugs.
The conversation on Science Friday provided a platform for Rovner to elaborate on the intricate processes involved in federal vaccine research and development. This included discussions on the significant investments being made in developing new vaccines, particularly in the wake of recent public health emergencies. The Biden administration, for instance, has continued to prioritize funding for biomedical research, including the National Institutes of Health (NIH) and the Biomedical Advanced Research and Development Authority (BARDA), which play crucial roles in supporting vaccine innovation. The focus often lies on platforms that can be rapidly adapted to address emerging infectious diseases, a lesson learned from the COVID-19 pandemic. Rovner likely delved into the complexities of clinical trials, regulatory approval pathways through the Food and Drug Administration (FDA), and the manufacturing and distribution challenges that accompany bringing new vaccines to the public. Data from the Centers for Disease Control and Prevention (CDC) consistently highlights the life-saving impact of vaccination programs, with vaccination rates for childhood illnesses remaining a key public health metric. However, discussions around vaccine hesitancy and the need for continued public education on vaccine safety and efficacy are also central to this discourse.
The discussion also likely touched upon the contentious issue of vaping, a public health concern that has escalated in recent years, particularly among young people. Rovner’s expertise would have been invaluable in explaining the current federal regulatory landscape, which has seen significant shifts. The FDA has been grappling with how to regulate e-cigarettes and other electronic nicotine delivery systems (ENDS). While the stated goal is to reduce youth initiation and addiction to nicotine, the industry has faced challenges in obtaining marketing authorization for its products. The FDA’s Tobacco Product Master Files (TPMFs) and Premarket Tobacco Applications (PMTAs) are central to this process. Data from the National Youth Tobacco Survey has repeatedly shown alarming rates of e-cigarette use among middle and high school students, prompting various interventions, including flavor bans and restrictions on marketing. Rovner’s analysis would have likely explored the tension between public health goals and the economic interests of the vaping industry, as well as the ongoing legal challenges and policy debates surrounding these products.
Furthermore, Rovner’s participation on Science Friday provided an opportunity to address the persistent problem of drug access and affordability. Despite various legislative efforts and policy discussions aimed at lowering prescription drug costs, many Americans continue to struggle to afford the medications they need. This issue is particularly acute for individuals with chronic conditions who rely on expensive, life-sustaining treatments. The Inflation Reduction Act of 2022, for example, included provisions for Medicare to negotiate drug prices for certain high-cost medications, a landmark policy change. However, the full impact of these provisions is still unfolding. Rovner’s commentary likely explored the various factors contributing to high drug prices, including patent protections, the complexity of the pharmaceutical supply chain, and the influence of lobbying efforts. Data from organizations like the Kaiser Family Foundation (KFF) consistently show that prescription drug costs are a significant financial burden for many households, leading to difficult choices between medication and other essential needs.
Rovner Addresses Supreme Court Decision on Mifepristone on NPR’s Morning Edition
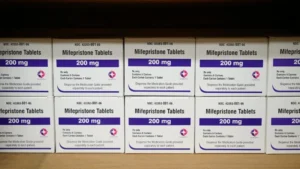
Adding to her busy week of public commentary, Julie Rovner also appeared on NPR’s Morning Edition on May 5 to discuss the Supreme Court’s handling of the legal challenges surrounding mifepristone, a medication used in medication abortions. This intervention by the nation’s highest court injected significant uncertainty into the availability of a widely used reproductive healthcare option, with profound implications for healthcare providers and patients across the country.
The Supreme Court’s decision, which ultimately preserved access to mifepristone while the legal challenges continue, was a critical moment in the ongoing debate over reproductive rights and access to medication abortion. Rovner’s expertise in navigating the complexities of health law and policy would have been instrumental in explaining the legal arguments presented to the court, the historical context of mifepristone’s approval by the FDA, and the potential ramifications of any ruling that restricted its availability. The legal battle centered on claims that the FDA had improperly approved and regulated the drug, despite its long history of safe and effective use. The Alliance for Hippocratic Medicine, a group of physicians, had filed the lawsuit, arguing that the drug poses significant health risks.
The timeline of events leading up to the Supreme Court’s involvement is crucial for understanding the context. Mifepristone was first approved by the FDA in 2000. Over the years, the FDA has modified its prescribing guidelines, including expanding access to the drug through telehealth and mail delivery, particularly in response to the COVID-19 pandemic. These changes became a focal point for legal challenges from opponents of abortion. In April 2023, a federal judge in Texas issued a ruling that would have suspended the FDA’s approval of mifepristone, a decision that was temporarily stayed by an appeals court. The Supreme Court’s intervention on May 5 prevented the Texas ruling from taking effect, allowing mifepristone to remain available under its existing FDA-approved conditions. However, the case was sent back to lower courts, indicating that the legal battle is far from over.
The implications of this ruling are far-reaching. For patients, continued access to mifepristone means that medication abortion remains an option in many parts of the country. However, the ongoing legal uncertainty could lead to confusion and barriers to access, especially in states with restrictive abortion laws. For healthcare providers, the decision brings some immediate relief, but the threat of future legal action remains a concern. The American College of Obstetricians and Gynecologists (ACOG) and other medical organizations have consistently affirmed the safety and efficacy of mifepristone, urging policymakers to rely on scientific evidence rather than political or legal challenges. The FDA itself has robust data demonstrating that mifepristone is safe and effective when used as prescribed, with serious complications being rare. This situation highlights the broader trend of legal challenges to reproductive healthcare services, often based on contested interpretations of scientific and medical evidence.
Gounder Discusses Rising Drug Prices on CBS News’ The Daily Report
Adding another layer of expert commentary to the national health policy discourse, Céline Gounder, KFF Health News’ editor-at-large for public health, appeared on CBS News’ The Daily Report on May 7. Her focus was on the persistent and often alarming rise in prescription drug prices, a topic that continues to be a major concern for patients and policymakers alike.
Gounder’s discussion likely addressed the ongoing debate surrounding the affordability of medications, even in the face of initiatives like "TrumpRx," a term often used to refer to various proposals and actions taken during the Trump administration aimed at lowering drug prices. Despite these efforts, data consistently indicates that the cost of prescription drugs in the United States remains significantly higher than in other developed nations. For example, studies by organizations like the RAND Corporation have shown that Americans pay substantially more for the same prescription drugs compared to citizens of countries like Canada, the United Kingdom, and Germany.
The underlying causes of high drug prices are complex and multifaceted. They include the lengthy and expensive process of drug development, which pharmaceutical companies argue justifies high prices to recoup their investments. However, critics point to factors such as the lack of price negotiation power for Medicare, patent evergreening (where companies make minor modifications to existing drugs to extend patent protection), and the absence of a robust competitive market for many essential medications. Gounder’s analysis likely provided context on these issues, explaining how the current system can lead to situations where life-saving drugs are out of reach for many.
The Biden administration has also made lowering drug costs a priority, with the aforementioned Inflation Reduction Act representing a significant legislative achievement in this area. The act allows Medicare to negotiate prices for a select number of high-cost drugs, a move that has been lauded by patient advocacy groups and criticized by the pharmaceutical industry. Gounder’s commentary on CBS News would have provided an important perspective on the effectiveness of these measures and the ongoing challenges that remain. Data from Medicare itself shows the potential for significant savings through negotiation, but the implementation is gradual, and the scope is currently limited to a small number of drugs.
The impact of high drug prices extends beyond individual financial strain. It can lead to patients rationing their medications, skipping doses, or foregoing treatment altogether, which can result in poorer health outcomes and increased healthcare utilization down the line. This creates a ripple effect throughout the healthcare system. Gounder’s role as an editor-at-large for public health underscores the broader societal implications of these pricing issues, highlighting how they affect the overall health and well-being of the population. Her insights on The Daily Report served to illuminate these critical issues for a broad audience, emphasizing the need for continued dialogue and policy innovation to ensure that essential medications are accessible and affordable for all Americans.
The consistent presence of KFF Health News experts on major national platforms underscores the organization’s vital role in shedding light on complex and impactful health policy developments. Through their informed analysis, Rovner and Gounder are contributing to a more informed public discourse, empowering citizens and policymakers to better understand and address the critical challenges facing the nation’s healthcare system.